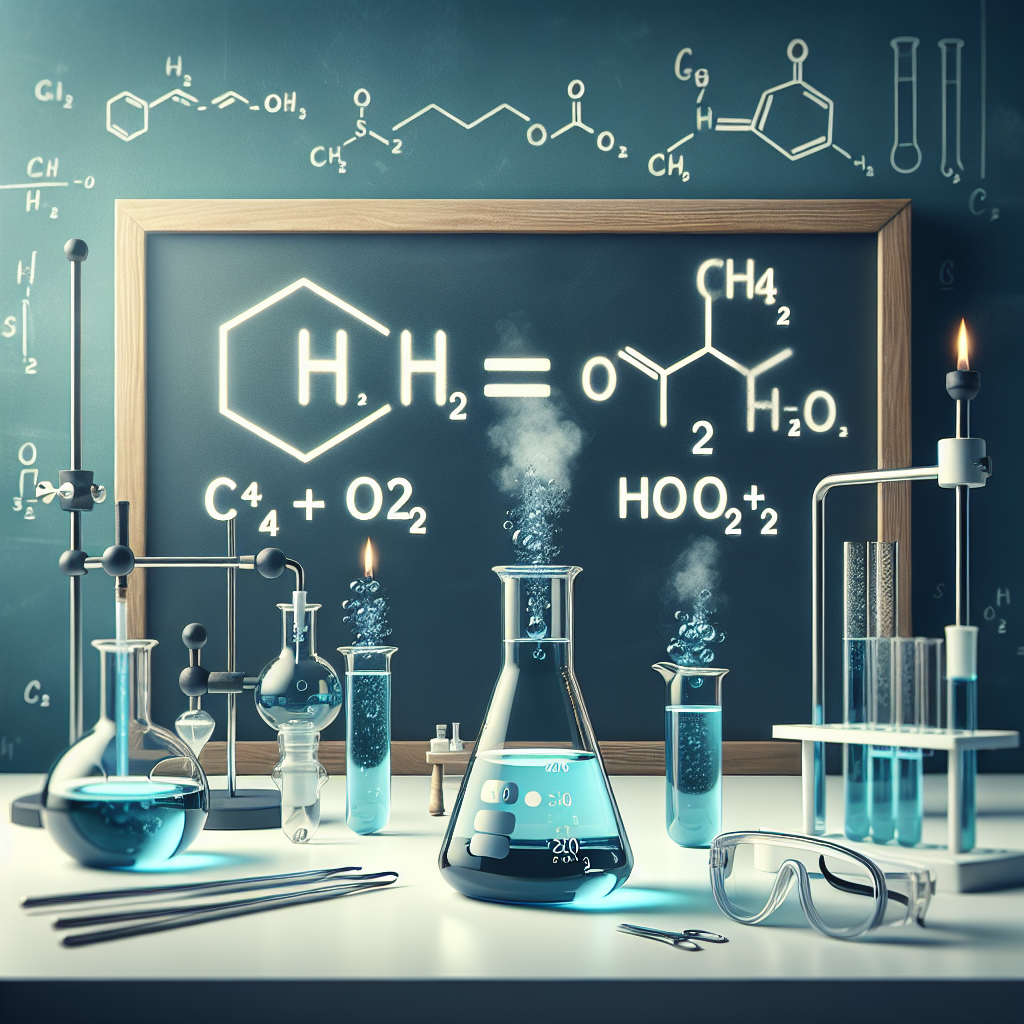
CH4 + 2O2 → CO2 + 2H2O
If you change the 2 in front of 2O2 to a 3, what will be the change in the results on the right side of the equation?
There are now 3H2O molecules. <- ?
Nothing changes in the equation.
There are now 3CO2 molecules. <- ?
There is an extra O2 molecule left over.
Simple Chemical Reactions Unit Test
1. There is an extra O2 molecule left over.
2. Covalent bonds tend to be less polar than ionic bonds.
3. PH3
4. replacement
5. Each carbon atom would share one pair of electrons with hydrogen and three pairs with the other carbon atom.
6. H2SO4 + 8HI → H2S + 4I2 + 4H2O
7. 2, 1, 2
8. Nitrogen and oxygen have an electronegativity difference of 0.5, so the bond is polar covalent, with oxygen pulling the electrons toward it.
9. Sodium donates an electron to chlorine.
10. selenium (Se)
11. bond type
12. six
13. K
14. indicator
the rest are essay questions sorry :/
yes, he’s correct. just took the test.
Yessirrr they are correct
my guy is a spooky potato
Potato: "the rest are essays, sorry"
Me: "NAAAH, POTATO U GUD U a life saver anyway"
He's 100% correct, B-T-dubs.
U took the test and you're sure these are correct right?
Thank youuuuuu
still correct
Does anyone know the essay questions?
(ALL CREDIT TO THE BRAINLY POST :>)
The bonding activity that occurs between phosphorus and chlorine is Covalent Bonding
Covalent bonding is an interaction between two atomic elements that share electrons between themselves.
In Phosphorus Pentachlorine PCl₅, covalent bonding occurs between Phosphorus and Chlorine.
This is because, Phosphorus is the central atom that is attached to 5 different chlorine atoms, each chlorine atom needs one electron each to complete its octet rule and since phosphorus has five valence electrons in its outermost shell, it shares it with each of the chlorine atoms.